

Scientific Contributionsįor more information about Rutherford’s scientific contributions, visit the Nobel Prize website. Rutherford had an enormous influence on the field of nuclear physics and mentored many future Nobel Prize winners and prominent scientists, including Chadwick, Niels Bohr and Otto Hahn. Their discovery of the atomic nucleus became an integral part of Rutherfords famous model of the atom we know and love today. Rutherford also coined the term “proton” and theorized about the existence of neutrons, which were discovered by his colleague and former student James Chadwick in 1932. In 1919, he became Cavendish Professor of Physics at Cambridge. The surprising results of this experiment ( Rutherford said, “It was as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you”) led Rutherford to formulate his model of the atomic nucleus, a revolutionary development in nuclear physics. Two years later, he, Hans Geiger, and Ernest Marsden conducted the Geiger-Marsden experiment, where they observed alpha particles scattering backwards when fired at a gold foil. In 1907, Rutherford returned to Great Britain to teach at the University of Manchester. In Montreal, he conducted the research that led to his Nobel Prize, including discovering the principle of radioactive half-lives and separating and naming alpha, beta, and gamma radiation. Thomson at the Cavendish Laboratory at Cambridge University, Rutherford became a professor and chair of the Physics Department at McGill University in Montreal, Canada. He is often called the “father of nuclear physics.”Īfter studying with J. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper.Ernest Rutherford (1871 – 1937) was a New Zealand-born British physicist and recipient of the 1908 Nobel Prize in Chemistry. In Bohr’s model the orbits of the electrons were explained by quantum mechanics. Niels Bohr built upon Rutherford’s model to make his own. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre. The plum pudding model was suggested as the first atomic model by J.J Thomson where he suggested that the atom was a sea of positive charge that surrounded small negative electrons. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. Later, he discovered the proton and in this sense, was the first to be able to split an atom. Robert Millikan was the first person to measure the elementary charge. The Rutherford model was devised by Ernest Rutherford to describe an atom. Bohr’s model explained why atoms only emit light.

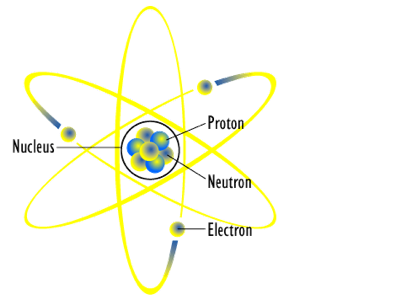
Electrons move around a nucleus, but only in prescribed orbits, and If electrons jump to a lower-energy orbit, the difference is sent out as radiation. He showed that radioactivity was the spontaneous breakdown of atoms, work he won the Nobel Prize for in 1908. In 1895, he became the first person to ever graduate with a Ph.D. In 1913, Niels Bohr proposed a theory for the hydrogen atom, based on quantum theory that some physical quantities only take discrete values. This meant that an electron circling the nucleus. But the Rutherford atomic model used classical physics and not quantum mechanics. Far from the nucleus are the negatively charged electrons. Most of the mass is in the nucleus, and the nucleus is positively charged.
That is, samples that have the same mass ratio are not necessarily the same substance. Ernest Rutherford (1871-1937) was an English nuclear physicist and is known as the father of nuclear physics. The Rutherford atomic model was correct in that the atom is mostly empty space. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general.
